Introduction:
Chimeric Antigen Receptor (CAR) T-cell therapies are remarkably efficient in treating B- and plasma-cell malignancies by targeting cell-of-origin antigens, eradicating both tumor cells as well as healthy B- and plasma-cell counterparts. Their loss can be compensated by immunoglobulin substitution until they regenerate from hematopoietic stem and progenitor cells (HSPCs). However, similar on-target, off-tumor cell-of-origin elimination would be detrimental in the context of HSPC-derived malignancies, such as acute myeloid leukemia (AML) or myelodysplastic neoplasia (MDS), as the CAR T-cell activity might lead to terminal ablation of hematopoiesis. In principle, CAR T-cells could be used to abrogate both, AML cells as well as HSPCs, followed by HSPC transplantation, ensuring hematopoietic recovery after termination of CAR T-cell activity and possibly without genotoxic preconditioning. This approach could be particularly beneficial for patients unable to tolerate radiation and high-dose chemotherapy.
In order to address this challenge, we developed adaptor-mediated CAR T-cells displaying the single-chain variable fragment E2, AdFITC(E2), that binds to fluorescein conjugated to antigen-binding adaptors in diabody format (Db-FM). Such tagged adaptors, used as single linkers or in combination, would then act as safety switch, modulating CAR T-cell activation, proliferation, and lytic activity.
Results:
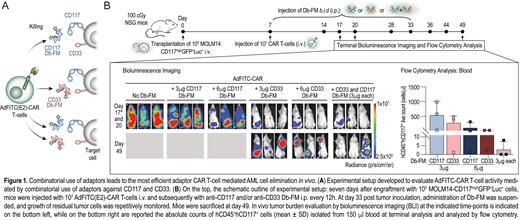
We generated second-generation AdFITC(E2)-CAR T-cells and multiple diabody-based adaptors against selected AML antigens. We then tested the diabodies targeting CD33 and CD117 with respect to their ability to mediate CAR T-cell biocidal activity as single adaptors or in combination. Combinatorial staining with CD117 Db-FM and CD33 Db-FM, each added at saturating concentration for the cognate antigen, resulted in higher fluorescein decoration of target cells, with more than two-fold increased fluorescence intensity detected by flow cytometry upon combined staining, thus enhancing AdFITC(E2)-CAR T-cell target density. In vitro cytotoxicity assays of healthy-donor-derived AdFITC(E2)-CAR T-cells, both against cell lines (MOLM14-CD117 +GFP +Luc +) as well as patient-derived AML blasts, revealed that dual adaptor use significantly improved tumor cell lysis compared to equimolar concentration of single adaptors. To further investigate the basis for increased killing by dual targeting, we performed live-cell imaging of effector-target cell interactions in microwells. The addition of both CD117 and CD33 Db-FM to the co-culture of MOLM14-CD117 +GFP +Luc + and AdFITC(E2)-CAR T-cells resulted in faster tumor cell lysis (measured by influx of PI) which correlated with a more rapid, durable engagement time of CAR T-cells with target cells. We next tested CAR T-cell biocidal activity in therapeutic xenogeneic mouse models engrafted with MOLM14-CD117 +GFP +Luc + where AdFITC(E2)-CAR T-cells in combination with CD117 or CD33 Db-FM administered as single adaptors were as efficient as direct CAR T-cells against the same antigens. Injection of AdFITC(E2)-CAR T-cells and both CD117 and CD33 Db-FM effectively inhibited tumor growth, outperforming monotherapies, and leading in some cases to full AML elimination in bone marrow and blood as shown by bioluminescence and terminal flow cytometry analysis (Figure 1).
Conclusions:
We here tested an adaptor CAR T-cell approach using multiple adaptors to on-off modulate and enhance CAR T-cell activity, focusing on AML as a relevant disease model with high clinical need for therapeutic improvement. The high heterogeneity in antigen expression on AML cells supports a combinatorial targeting strategy, individualized based on the respective AML immunophenotype. Due to their relatively small molecular weight (~55kDa), diabody-based adaptors are rapidly cleared from the body via the kidneys allowing for rapid control over AdFITC-CAR T-cell on-off activity prior to HSPC transplantation. We envision that this approach has the clinical potential to improve CAR T-cell safety profiles by enhancing the specificity towards target cells, while ameliorating the side-effects against healthy tissues expressing only one antigen of the combination.
Disclosures
Myburgh:ATLyphe: Current Employment. De Luca:Philochem AG: Current Employment. Favalli:Philochem AG: Current Employment. Neri:Philogen S.p.A.: Current Employment, Current equity holder in publicly-traded company, Divested equity in a private or publicly-traded company in the past 24 months, Membership on an entity's Board of Directors or advisory committees.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal